
1 ppm represents a concentration of 1 mg in every 1 kg of toothpaste. This is incorrect right? We need to take into account the vapor pressure of formaldehyde at the elevated batch temperatures using clausius clapeyron eq or antoine's parameters right ? Am I missing something here? Please look at picture and I can provide more info as well. The concentration of NaF can be expressed in parts per million (ppm). 3 lbs and dividing it by a rough estimate of the volume of the mixer. For example, a sample with a mass concentration of 100 mg/l will have.

She calculated that we were generating a concentration of formaldehyde by taking the mass of formaldehyde in the batch (solid/liquid mass) of. To convert from ppm by mass to ppm by volume, divide by the density of the particles. Now the picture attached is our industrial hygienist way of calculating ppm. Question: We are trying to calculate how much formaldehyde is generated in ppm during the batch to see if our respirators can handle it and to also get an LEL %. This measurement is the mass of a chemical or. The batch mixes until it reaches 350F and then we dump the batch. This is an abbreviation for parts per million and it also can be expressed as milligrams per liter (mg/L). Example: Concentration of stock solution: 1000 ppm (Concentration A) New standard: 10 ppm (Concentration B) Volumetric flask: 100 mL (Volume B) So, for this example, to make a 10 ppm standard, add 1 mL of the 1000 ppm standard to a 100 mL volumetric flask. Its a butyl rubber based batch mixed in a banbury mixer so a lot of heat is generated from the rubber masticating. %=mg/l× 22.Have a batch with a 3 lb curative package. It is generally used for measuring concentrations in liquids, and only rarely for gaseous concentrations.Ĭonverting from one unit of concentration to another % → ppm This unit expresses the concentration in one litre of air (1000mL) of a substance in terms of its mass (measured in milligrams).
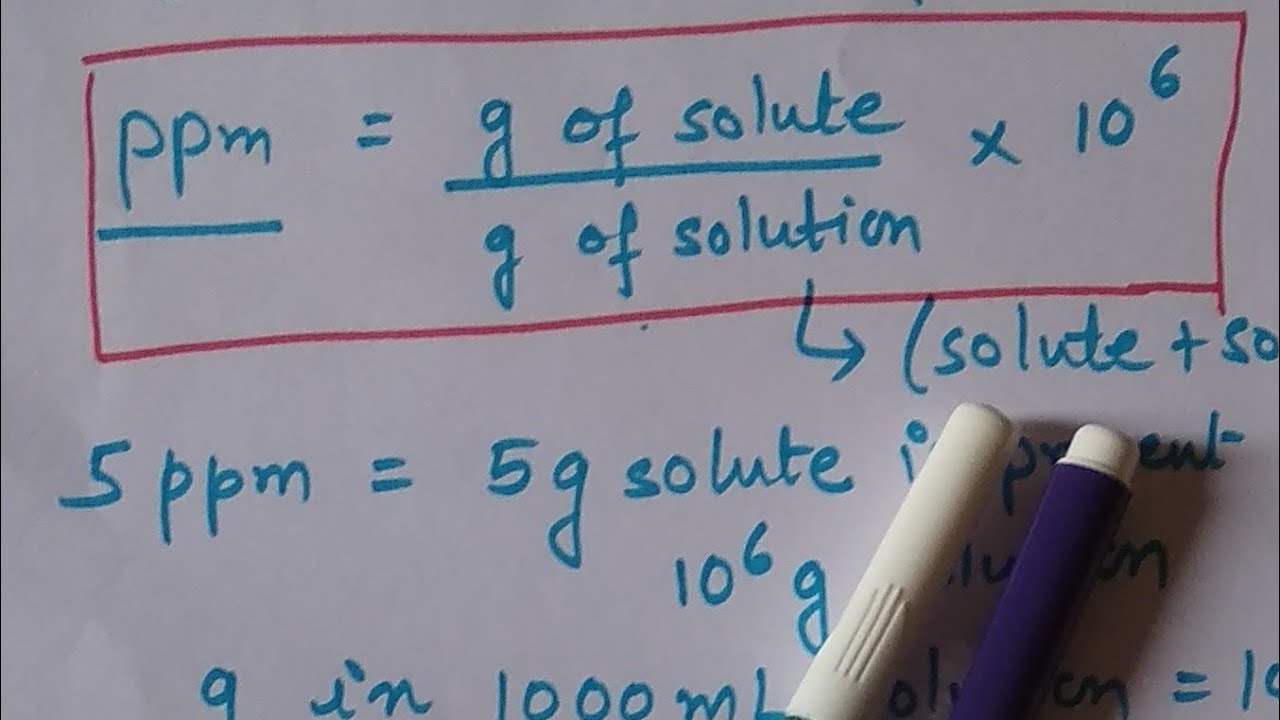
Sometimes, however, the gas concentration is expressed as the mass concentration in milligrams per cubic meter (mg/m3). By definition, it is the number of gas particles per million air particles, 1 ppm 1/1000000 0.0001 0.001. Parts per million by mass is equivalent to milligrams per liter (mg/L). It is primarily used for particle-like substances, and only rarely for gaseous concentrations. Ppm is often used for low concentration values, typical for air pollution. A measurement of concentration on a weight or volume basis. This unit expresses the concentration in one cubic metre of air (equivalent to 1L or 1000mL) of a substance in terms of its mass (measured in milligrams). To convert between units just type the know value. This unit that expresses concentration in parts per million is measured as the volume (denoted in litres ) of a substance found in 1L of a medium such as air. Online Conversion between ppm (parts per million), ppb (parts per billion) and percent concentration. This unit expresses concentration in parts per hundred (percentage) of a substance in 100mL of a medium such as air. To convert ozone concentration in oxygen to another unit of measure use the calculator below. Mg/L Units for measuring concentration Common units for measuring concentration % (percent)
